A new way to design for additive orthopedic implants

Written by nTop
Published on May 6, 2020
Designing for additive medical devices is only painful when you're not properly equipped to do the job. What if you could use a design software that gives you the tools you need to not only keep up with additive, but push the limit of its capabilities?
More than once I’ve been told by orthopedic device companies from around the world that they wish to expand their business beyond their own borders. In this globalized economy, it only makes sense to take that leap into a brand new market. The usual next step for these products is the highly desired European CE marking, but the holy grail for medical devices has always been the coveted FDA clearance. Why? The process to clear an implant in the United States can be one of the most arduous in the world. With a highly scrutinous regulatory body and a thoroughly demanding submission process, it’s not easy on the wallet either; simply getting past this hurdle is an accomplishment in and of itself.
So when 3D printed medical devices became the flavor of this past decade, the race was on to be the first to market with a cool new design using this hot new process. The only problem? The technology was quickly outpacing regulation and the FDA was scrambling to properly determine exactly what the rules should be. It wasn’t until the tail end of 2017 that the main guidance document released its final version. And for the numerous 3D printing companies that had sprung up overnight to serve this potentially lucrative market, this was the only real guidance they had.
Fast forward to today, the submission process is a bit more understood as quite a few additive orthopedic devices have had their 510(k) Premarket Notification cleared. The awareness of additive technology has slowly become more and more ubiquitous within the industry, and companies are able to leverage an increasing number of predicates for their own submissions. However, even as the manufacturing process itself begins to mature, it is becoming apparent that the biggest thing holding the industry back is the software.
Design limitations
CAD tools are the cornerstone of digital design. Parametric solid modeling and surfacing have been the standard for as long as many engineers can remember, and to give credit where credit is due, it has done quite a fine job propelling the engineer far beyond the limitations of drafting by hand. Unfortunately, in its attempt to take drafting to the next level, CAD has also adopted its limitations; limitations that didn’t reveal themselves until additive reared its head as the future of manufacturing.
In the past decade, 3D printing has enabled design in the likes of which the world has never seen. Lattice structures were unthinkable in a time when manufacturability was defined by only what the tool could see. Contoured and organic shapes used to demand a high level of surfacing skill, yet makers and hobbyists today can pump out a “topology optimized” bookshelf bracket with barely a second thought. But as manufacturing continues to move forward, are digital design tools keeping pace? You can laboriously create a complex lattice structure in your traditional CAD tool, but it begins to beg the question: does it even make sense to?
Enter nTop. As a relatively young company amidst the presence CAD giants, nTop Platform is a design software solution that pushes the capabilities of additive manufacturing instead of simply keeping up with them. The modeling technology behind nTop Platform is not just different, it is a uniquely efficient approach to designing and managing the complex geometry that has dominated the manufacturing industry, especially in medical devices.
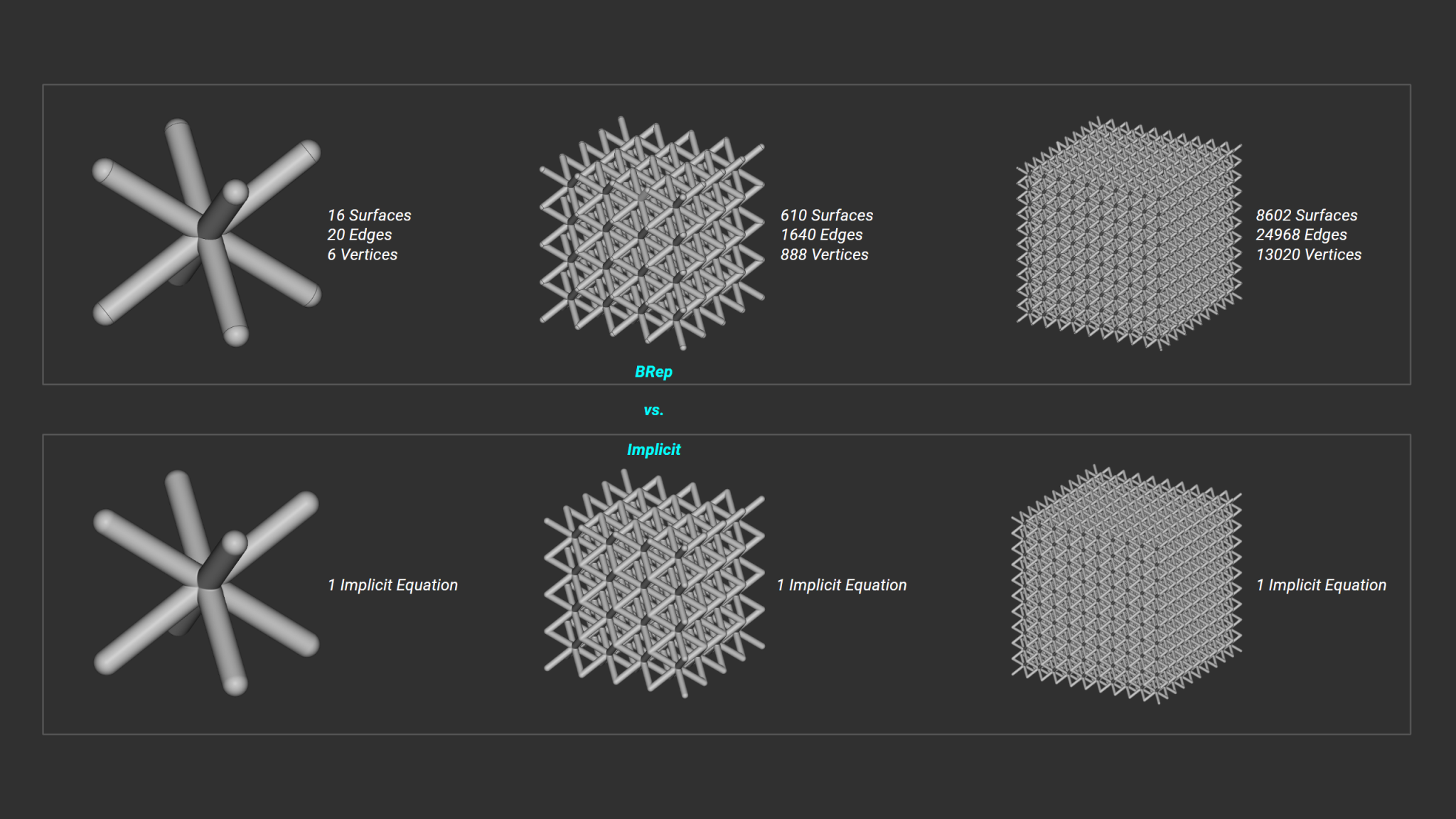
When people think of 3D printed orthopedics, they often think LimaCorporate’s Delta TT acetabular cup or Stryker’s Tritanium spinal cages. These were groundbreaking additive products that have launched wave after wave of comparable orthopedic designs leveraging ordered and randomized lattice structures. Sadly, what also rode along these waves were the millions of triangle elements and heavy file sizes when defining these lattice structures using common printable formats.
Generating these lattices potentially meant spending a solid hour watching a loading bar slug its way from one side to the other. And if you had an entire part family to go through, you’d spend the better part of your week simply twiddling your thumbs. What made matters worse were the irreversibility of some of the lattice tools available at the time. If an upstream design change happened to be necessary, all of your lattice structures needed to be regenerated from scratch.
The usability just wasn’t there.
Putting it all together
A highly scrutinous regulatory body, a modeling technology that can’t keep up, and a design process that just doesn’t make sense. How does one solve all of these problems?
nTop is looking at medical device design in a whole new way. We don’t just see design as a means to an end; we value the design process just as much as the completed output. In nTop, design engineers are not just creating a part, they are developing a workflow. With workflow-oriented design, we are focused on ensuring the user can design with four critical aspects in mind.
Creating a traceable, digital trail that captures all the input parameters that define your device, enforcing product credibility and mitigating development risk. Designing workflows that can be repeatable and reusable across part families and product lines, significantly reducing the development cycle. Collaborating through modular workflow architecture and distributable design intent.Leveraging efficient and effective design tools to develop next-generation osseointegrative interfaces and patient-specific devices.
Closing
In the coming weeks, we will dive into each of these aspects as they all play a crucial role in the product development process of medical devices. We look forward to sharing with you how nTop has taken each of them into consideration as we continue to push the industry with cutting edge design software.
Looking for more on medical devices? Check out our webinar on Advanced Workflows for Orthopedic Device Design! In this session, I co-host with Matt Shomper of Tangible Solutions to talk about biologically relevant structures and showcase a unique data-driven design process.

nTop
nTop (formerly nTopology) was founded in 2015 with the belief that engineers’ ability to innovate shouldn’t be limited by their design software. Built on proprietary technologies that upend the constraints of traditional CAD software while integrating seamlessly into existing processes, nTop allows designers in every industry to create complex geometries, optimize instantaneously, and automate workflows to develop breakthrough parts and systems in record time.
Related content
- VIDEO
Design better implants for osseointegration with the Lattice Pore Size Block

- VIDEO
nTop CDS 2024: Exploring the cutting edge of computational design with Matthew Shomper

- CASE STUDY
Cobra Golf designed their LIMIT3D irons 50% faster with nTop

- GUIDE
Download: Advanced design software and additive manufacturing for personalized implants

- GUIDE
Download: Design Automation for Medical Devices
